The Complexity Of Microbiome Data
In addition, the genetic architecture, the landscape of contributions of host genetics to a given microbiome phenotype, is at best poorly understood although studies of hostgenetic interactions with microbiome suggests polygenicity. Moreover, the reported percentage of microbiome variation explained by the associated alleles are generally very small , and thus unable to explain much of the variability in microbiome phenotype. Given this effect size, large sample sizes will be required to detect modestly associated variants. Meanwhile, the high level of trait collinearity coupled with complex correlation structure also make it challenging for statistical methods. Even though parametric linear models remain the cornerstone for genetic association studies and have played pivotal role in mGWAS carried out to date, they are limited to detecting non-linear interaction patterns. In particular, when modeling complex structures such as varying effect and non-linear interactions, the exponential rise in the number of parameters increases computational cost and reduces statistical power . Moreover, linear models generally treat interaction effects as factors with independent marginal effects a strategy that lowers its power in the presence of interaction effects . Given these limitations, there is a need to develop mGWAS-adapted statistical methods to complement existing linear models.
Phylogenetic Analysis Of Microbial Community
3.1.1 Culture-dependent methods
3.1.2 Culturomics
The significance of culture-dependent methods cannot be undermined for the identification of microbes from the gut microbial community. Therefore, microbiologists have rediscovered and focused once again to revive culture-based methods by adding many sophisticated instrumentations and suitable growth media. This has allowed growing most of the unculturable bacteria that were earlier thought to be impossible in a lab environment. Hence, it will allow to know more about the functional aspects of gut microbiome that include its composition, microbial gene expression, metabolic pathways and host-bacteria relationships . Actually, diverse types of favorable growing and incubation conditions are required to grow unculturable microbes that are provided by the new culturomics procedures. Currently, more than 50% of bacterial species that were earlier identified by classical 16S rRNA metagenomics could be re-identified with the help of culturomics. Simultaneously, it will also allow isolating hundreds of new bacterial species in the gut microbial ecosystem in the near future .
3.1.3 Microfluidics assays
Human Genetics And Immune Interactions In Early Development
The composition of the human microbiome is unique in each individual, and the differences among individuals are large compared to the typical biochemical differences within a person over time,. Identical twins are barely more similar to one another in microbial composition and structure than are non-identical twins, at least over the range of environmental factors captured in studies to date, suggesting that the effect of the human genome is limited, and that most of microbial community assembly may be determined by environmental factors. Early underpowered studies suggested that monozygotic twins were no more similar in terms of their overall gut microbiota than dizygotic twinsâ, although larger cohort sizes show a small but statistically significant effect of genetics on microbiome composition in twins with certain taxa being identified as highly heritable, such as Christensenella. However, one way to rationalize this is that the number of bacteria that are able to successfully colonize humans is limited. Colonizing initially germ-free mice with diverse environmental samples demonstrates that very few bacteria present in the environment can survive in the mouse gut, and those that do are rapidly displaced by human- or mouse-derived bacteria on exposure Furthermore, human immune responses shape responses to changes in the microbiome and are involved in shaping the microbiome itself.
You May Like: Do Bananas Help With Bloating
Why Is The Microbiome Important
The trillions of bacteria in your gut have a profound influence on our health, metabolism, and even disease protection.
When we hear the words microbe, bacteria, or virus, we tend to think of something bad, but not all of these microorganisms cause disease. In fact, we rely on them to perform the functions we may not be able to.
A balanced microbiota with beneficial and probiotic bacteria performs many other functions, like maintaining the correct pH to deter pathogens and keeping the gut lining healthy. Research even shows that gut microbes are involved in maintaining a healthy body weight.
Healthy And Diseased Microbiomes
Much like the Human Genome Project was created to catalog the entire sequence of our own DNA, the Human Microbiome Project worked to catalog the genetic information of the microbes in and on the human body that contribute to our survival. By looking at the genetic variation of microbes at these different body sites, as well as among different individuals, scientists hope to understand more about how our microbiome keeps us healthy or may contribute to disease.
Additionally, a group of Washington University researchers worked with the McDonnell Genome Institute to sequence the microbiomes of certain diseased patients including those with Crohns disease children with fever of unknown origin and an often fatal condition in premature babies called Necrotizing Enterocolitis, that causes inflammation of the intestinal tissue. The McDonnell Genome Institute also collaborated with researchers at other institutions who looked at how the microbiome is associated with sexually transmitted diseases and the male adolescent urethra, as well as a project dealing with the skin microbiome as it relates to acne.
These demonstration projects were started to help guide the direction of future clinical research by looking at the potential links between the makeup of our microbiomes and specific disease states. This research could ultimately provide doctors with new diagnostic and therapeutic approaches for a number of diseases.
Read Also: Are Probiotics Good For Colitis
The Knowns And Unknowns Of The Human Microbiome
Are you aware that the unculturable fraction of the microbiome may account for 80% of the observable bacteria? Here, Andrew Maltez Thomas and Nicola Segata from the University of Trento scope out the unknowns in contemporary human microbiome research.
Traditionally, microbial communities that inhabit the human body and surrounding environments have been characterized through culturing on selective plates. However, this approach shows a low sensitivity and does not allow for exploring the unculturable fraction of the microbiome that may account for between 60% and 80% of the observable bacteria.
Although culture-independent methods based on characterizing the 16S ribosomal ribonucleic acid genes and shotgun sequencing of the whole genome have been developed, understanding the complexity of the microbiome in human and non-human environments remains a distant target.
In an attempt to further our knowledge, Andrew Maltez Thomas and Nicola Segata from the University of Trento scope out the unknowns in contemporary human microbiome research.
However, the diversity of a large fraction of the human microbiome remains unexplored.
Last, but not least, it is difficult to have a complete picture of the functional diversity potential of the human microbiome, mainly due to the inherent nature of high-throughput methodologies that show an imbalanced cost-effective relationship. According to the authors, this is probably the most important caveat in the field.
Reference:
Microbial Metabolites Influence Physiology
A large number of extrinsic and intrinsic factors, including diet, antimicrobials and immunity influence human microbiomes, in particular the gut microbiome which houses the largest number and diversity of microbes. In turn, the bioactive products of the microbiome shape human cell function locally, and at remote sites. In studies of mono-colonized GF mice, members of the intestinal microbiome were shown to strongly influence mammalian energy harvest and metabolism, and produce a suite of microbial-specific metabolites in physiologically relevant concentrations, a large number of which enter the circulation. Thus, microbial activities at one body habitat may influence physiological conditions and cell function at a remote site. For example, increased concentrations of trimethylamine N-oxide associate with arthrosclerosis and is dependent on gut bacterial metabolism of dietary phosphatiylcholine,, providing the first evidence for a diet by gut microbial interaction governing cardiovascular disease.
Read Also: Bananas Make Me Bloated
What Does Our Microbiome Do
Lots. The gut microbiome controls the storage of fat and assists in activating the genes in human cells involved with absorbing nutrients, breaking down toxins and creating blood vessels.
These helpful microorganisms replenish the linings of the gut and skin, replacing damaged and dying cells with new ones. Equally vital is their role in preventing illness.
Our native microbes compete with invading ones, preventing them from getting a foothold. Were born with an immune defence system only partially formed.
Its the interaction with microbes that shapes it, influencing the classes of immune cells that are generated and the development of the organs that make and store them.
Spoiler Alert: Its A Lot According To An Early Study
There are likely more genes in the body than there are stars in the universe.
US scientists have begun the daunting task of trying to work out how many genes there are in the human microbiome.
Even when you consider just the gut and the mouth the numbers are potentially overwhelming.
Microbiologists and bioinformaticians from Harvard Medical School and Joslin Diabetes Centre gathered all publicly available sequencing data on human oral and gut microbiomes and analyzed the DNA from around 3500 samples 1400 from mouths and 2100 from guts.
In all, there were nearly 46 million bacterial genes 24 million in the oral microbiome and 22 million in the gut.
That leads the team to suggest that there may be more genes in the collective human microbiome than there are stars in the observable universe and that at least half of them may be unique to each individual.
Ours is a gateway study, the first step on a what will likely be a long journey toward understanding how differences in gene content drive microbial behaviour and modify disease risk, says Harvards Braden Tierney, first author of a paper in the journal Cell Host & Microbe, with admirable understatement.
Most research to date has focused on mapping the types of bacteria that inhabit our bodies in an effort to determine whether and how the presence of a given bacterial species might affect disease risk, the researchers say.
Read Also: Probiotics Align Ingredients
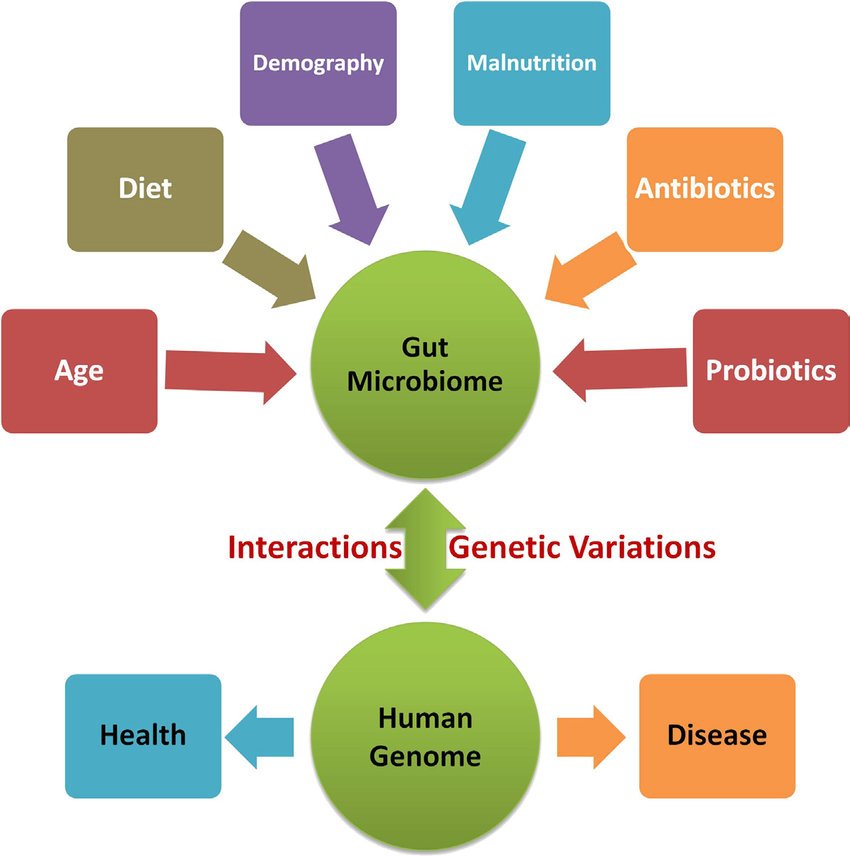
Factors Influencing The Human Microbiome
To alter the microbiome deliberately for preventive or therapeutic purposes, or use it to understand a particular medical condition, we must first understand the factors that influence its composition. We have reviewed many of these factors in detail recently,, so we provide only a brief summary here.
In A First Scientists Map The Genetic Diversity Of Microbes Residing In The Human Gut And Mouth
How many stars are there in the observable universe? It was once deemed an impossible question, but astronomers have gleaned an answerabout one billion trillion of them.
Now, scientists at Harvard Medical School and Joslin Diabetes Center have embarked on what could be a similarly daunting quest: How many genes are there in the human microbiome?
In a study published Aug. 14 in the journal Cell Host & Microbe, a team of microbiologists and bioinformaticians offer a first glimpse of the array of genes that make up the bacterial universe residing in each of us.
The findings thus far: There may be more genes in the collective human microbiome than stars in the observable universe, and at least half of these genes appear to be unique to each individuala diversity far exceeding the researchers expectations.
The research is believed to be the largest analysis of its kind to date and the first one to include DNA samples from bacteria that reside both in the mouth and the gut. Past studies have focused on one or the other.
Even so, the work marks only the beginning of efforts to analyze the entire genome of the human microbiome.
Ours is a gateway study, the first step on a what will likely be a long journey toward understanding how differences in gene content drive microbial behavior and modify disease risk, said study first author Braden Tierney, a graduate student at Harvard Medical School.
A microbes evolutionary organ
You May Like: Does Banana Cause Bloating
Towards Mechanistic Studies Of The Microbiome
Mechanistic studies of the microbiome are typically difficult to perform in humans, in part because of tremendous genetic and lifestyle heterogeneity, and there are ethical issues associated with colonizing human subjects with microbes that are hypothesized to cause disease. Therefore, most of what we know currently stems from experiments in animal models. However, recent studies that complement observations in humans with interventions in animal models have produced striking new insight into the microbial origins of disease that cannot be acquired from human studies alone.
Microbial Associations With Host Genome Variants Are Body Site
Finally, we assessed associations between host genome and microbiome variation in a non-targeted manner directly through a genome-wide association study. We performed the analysis separately for each body site, concentrating on SNVs with MAF> 0.1 and comparing them to both microbial species- and metabolic pathway-level abundances. We used ordinary linear regression models, taking into account the effects of sex, ethnicity, and sample collection location. After filtering the microbial features , we identified 120160 species in GI tract and oral samples and approximately 25 species in skin and vaginal samples. The number of metabolic pathways passing filtering was considerably higher, between 350 and 530 major pathways per site. Together the large numbers of SNVs, body sites, and microbial features in the analysis impose a strict significance criterion , which, in combination with our modest sample size, limits our discovery potential to associations with very large effect sizes.
We next ran the association analysis on all common SNVs. We did not see any associations with p values smaller than multiple testing-corrected significance limits. However, there were a number of associations with relatively small p values. These associations form a rich source of information for future microbiome-wide association studies and investigators interested in specific genes or microbial species.
You May Like: Why Does Lettuce Give Me Diarrhea
Factors Shaping The Gi Microbiota
The microbiota composition is subject to shaping by host and environmental selective pressures. To protect from injury and maintain homeostasis, the GI tract limits exposure of the host immune system to the microbiota by recruitment of a multifactorial and dynamic intestinal barrier. The barrier comprises several integrated components including physical , biochemical and immunological factors . An individual microbe’s longevity is determined by whether it is contributing to the range of essential functions on which host fitness relies. It is proposed that organisms who do not contribute beneficial functions are controlled by, and may occasionally be purged during, for example, transferral of the microbiota to a new host .
Gut microbes must be adapted to a certain type of lifestyle due to the relatively fewer number of biochemical niches available in the gut, compared with other microbial-rich environments. In the gut, energy can generally be derived through processes such as fermentation and sulphate reduction of dietary and host carbohydrates. The organisms that can survive in the gut are therefore limited by their phenotypic traits .
The availability of sulphated compounds in the colon, either of inorganic or organic origin, can influence specific groups of bacteria such as sulphate-reducing bacteria, which are residents of the gut microbiota that have been implicated in the aetiology of intestinal disorders such as IBD, IBS or colorectal cancer .
Importance Of The Human Microbiome Project
The human microbiome makes up about one to two percent of the body mass of an adult. It has been likened to a body organ. But, unlike say a heart or a liver, the importance and function of the microbiome is just starting to be appreciated.
It has long been known that bacteria are involved in certain body processes, such as digesting food and producing vitamins, but the microbiome appears have a much broader impact on our health than was previously realized. The community of microbes in an individual may influence the susceptibility to certain infectious diseases, as well as contribute to disorders such as obesity and diabetes. It may also contribute to the development of some chronic illnesses of the gastrointestinal system such as Crohn’s disease and irritable bowel syndrome. Some collections of microbes can determine how one responds to a particular drug treatment. The microbiome of the mother may even affect the health of her children.
A more complete understanding of the diversity of microbes that make up the human microbiome could lead to novel therapies. For example, it may be possible to treat a bacterial infection caused by a “bad” bacterial species by promoting the growth of the “good” bacteria. Microbiome transplants are already being used to combat certain illnesses, such as Clostridium difficile infections, to establish more healthful bacterial populations.
You May Like: Align Digestive Care Side Effects
Bacterial Components Of A Healthy Microbiome
Fig. 1
Although less well-studied than the gut, many other body habitats within healthy individuals are occupied by microbial communities . Community composition is more similar within than between habitats , although, in turn, inter-individual differences within habitats are much greater than intra-individual variability over time . Oral sites harbor particularly diverse microbiomes , similar in complexity to the microbiome of the gut , and tend to be dominated by Streptococcus spp. . Skin sites differ primarily with the local properties of the skin and are colonized primarily by Corynebacterium, Propionibacterium, and Staphylococcus . The healthy vagina contains one of the most remarkably structured microbial ecosystems, with at least five reproducible community types, or community state types, each dominated by a single species of Lactobacillus or by a mixture of other microbes including Gardnerella . Significant determinants of a womans community state type include race/ethnicity and pregnancy , although even in this structured ecosystem within-subject longitudinal variation is substantial and, to date, has no fully explained causes.
Our Evolving Understanding Of The Healthy Microbiome
Early studies sought to identify the normal set of microbes that colonize healthy people, primarily in the gut, by culture and characterization of physiological properties. Such studies best highlight organisms that grow well in the lab environment, such as Escherichia coli. This bias led to the perception that E. coli is an abundant and prevalent member of the human gut microbiome . The introduction of strictly anaerobic techniques in the 1970s allowed the recovery of more than 300 bacterial species from the gut alone furthermore, the counting of viable cells within standardized serial dilutions in selective media permitted quantification of these species. A summary of four large studies from this era looking at stool samples from 141 Americans on different diets found that bacteria of the genus Bacteroides and anaerobic cocci were both prevalent and abundant, whereas the genus Clostridium was ubiquitous in lower abundance, though no single species was observed in all subjects. Other prevalent but lower-abundance bacteria included members of the genera Bifidobacterium, Eubacterium, Lactobacillus, and Streptococcus, as well as facultative anaerobes such as Escherichia.
You May Like: What Can Cause Bloating And Shortness Of Breath