Immunity In The Lungs
The gut microbiota can aid the clearance of pathogens by enhancing the innate immune responses to pathogens in the lungs. In a study designed to investigate the role of commensal microflora in the gut on the host defense in pneumonia through toll-like receptors , the authors found that antibiotic pretreatment to deplete commensal gut flora prior to Escherichia coli pneumonia challenge led to increased bacterial burdens in both the blood and the lungs. They also noted cytokine suppression as well as suppressed nuclear factor B activity in the intestine. They concluded that the gut microbiota is critical in inducing TLR4 expression and nuclear factor B activation of intestinal and lung innate immune defense against E. coli pneumonia . Bacterial Nod-like receptor ligands including a NOD1 ligand, MurNAcTriDAP, and a NOD2 ligand, muramyl dipeptide, from the gastrointestinal tract have also been shown to rescue host defenses in the lung . Additionally, gut microbiota promote lung immunity following vaccination . Cell wall components and flagellin of gut bacteria have been shown to stimulate TLRs for effective adaptive immune responses to influenza .
The Lung Microbiome Associated With Established Asthma And Atopy In Adults
The respiratory microbiome in adults with established asthma has a lower bacterial diversity compared to healthy subjects and an increased richness, both correlating to asthma severity . Several groups have reported an increased abundance of the phylum Proteobacteria, and in particular the genus Haemophilus in asthmatics . Additionally, Huang and colleagues found that higher abundance of Proteobacteria was related to lower asthma control and asthma exacerbations which was accompanied by the induction of Th17-related genes . Especially, enrichment of Haemophilus and Moraxella, both belonging to the class of Gammaproteobacteria, were associated with severe airway obstruction and airway neutrophilia . Most of the mechanistic studies on the influence of airway microbiome on asthma development were conducted in murine models . One of the few mechanistic approaches in human adult asthma related the finding of increased Haemophilus parainfluenzae with in vitro studies . The authors could demonstrate that Haemophilus parainfluenzae was able to activate Toll-like receptor 4, which subsequently led to transcription of pro-inflammatory factors such as IL-8 and at the same time inhibited corticosteroid-related pathways. The induction of corticosteroid resistance is an important factor as corticosteroid treatment is one of the mainstay treatments in asthma and inflammation.
Gut Microbiota And Local Immunity
Gut microbiota effects on the local immune system have been extensively reviewed . Briefly, the gut microbiota closely interacts with the mucosal immune system using both pro-inflammatory and regulatory signals . It also influences neutrophil responses, modulating their ability to extravasate from blood . Toll-like receptor signaling is essential for microbiota-driven myelopoiesis and exerts a neonatal selection shaping the gut microbiota with long-term consequences . Moreover, the gut microbiota communicates with and influences immune cells expressing TLR or GPR41/43 by means of microbial associated molecular patterns or short-chain fatty acids . Data focused on the gut mycobiota’s impact on the immune system are sparser. Commensal fungi seem to reinforce bacterial protective benefits on both local and systemic immunity, with a specific role for mannans, a highly conserved fungal wall component. Moreover, fungi are able to produce SCFAs . Therefore, gut mycobiota perturbations could be as deleterious as bacteriobiota ones .
Recommended Reading: Salad Causing Diarrhea
As Unique As Your Fingerprint
The lung microbiome is analysed by taking sputum samples from individuals. Modern laboratory techniques RNA and metagenomic sequencing) have enabled us to gain information about the quantity and identity of the different bacteria present in a sample.
The composition of the lung microbiome is influenced by many factors and is unique to an individual. Diet, exercise, antibiotic use, ethnicity, environmental factors, ageing and life events all have an influence and cause it to change over time.
The three main factors that influence the lung microbiome are:
Proactively Maintain A Good Balance Of Microbes In The Mouth With The Help Of Oral Supplements Tongue Scraping And Microbially
- I use the new oral spray MegaCidin, which combines herbal extracts from oregano, grapeseed, black walnut, tea tree, echinacea and others, with the bacillus spore probiotics Bacillus subtilis and Bacillus coagulans to support a normal oral microbiome.
- Dentalcidin is a supportive toothpaste containing similar microbe-balancing ingredients.
- For more pronounced oral imbalances, the liposomal rinse Dentalcidin LS can help penetrate the harder to reach nooks and crannies of the oral mucosa and gums for more complete support.
- Scraping the tongue daily with an antimicrobial copper tongue scraper can promote fresh breath, and help to balance the microbial environment of the mouth. You can also scrape the inside of your cheeks. The copper scraper is naturally antimicrobial and can be used over and over again.
You May Like: Probiotic For Colitis
Future Respiratory Microbiome Research
From the above discussion, participants in the workshop agreed on the following nine specific aspects that need to be specifically addressed by future respiratory microbiome research:
Normality patterns: Studies performed in healthy subjects so far have clearly demonstrated that there is a rich microbiota in the respiratory system that includes microorganisms from the Firmicutes, Bacteroidetes and Proteobacteria phyla, and displays a close similarity to that of the oropharyngeal microbiota. Normality patterns for viruses and fungi still need to be defined, however. The microbial composition of the respiratory microbiota changes in chronic respiratory diseases, but the timing and the distribution of these changes are only partially known.
Standardisation: There is a pressing need to standardise protocols to be used to analyse the respiratory microbiome, including sampling, processing and bioinformatics methodologies. The creation of consortia and networks for research on this topic would facilitate this standardisation and, as a result, the possibility of sharing results from different cohorts.
Non-cultivable and/or non-pathogenic bacteria: 16S rRNA gene analyses have shown high relative abundance and specific patterns of non-cultivable microorganisms in bronchial and lung samples obtained from patients with COPD, IPF, CF and bronchiectasis. The role of specific species previously considered non-pathogenic needs to be addressed in these different clinical conditions.
It’s Not Just Bacteria Is It
We’re colonized with a complex mix of bacteria, fungi, protozoans, helminths and viruses, yet the majority of microbiome studies worldwide only include bacteria. What influence do non-bacterial microbes have in the gut microbiome? How do these microbes affect bacterial communities? How to they interact with our immune system? We’re applying ecological theory concepts to explore the relationships between microbial eukaryotes and prokaryotes, using bioinformatic and mouse model strategies.
Also Check: Does Almond Milk Cause Bloating
Maintain Good Bile Flow
Support bile flow with srategies such asintermittent fasting, or a supportive supplement such as MegaGuard, Milk Thistle, or an herbal combination such as Lipotropic Complex by Integrative Therapeutics. Bile acids are antimicrobial in nature and bile flow can get sludgy with SIBO, leaky gut, and gastrointestinal overwork and inflammation. You can also supplement with bile acid salts, but intake levels can be touchy as excess can cause diarrhea.
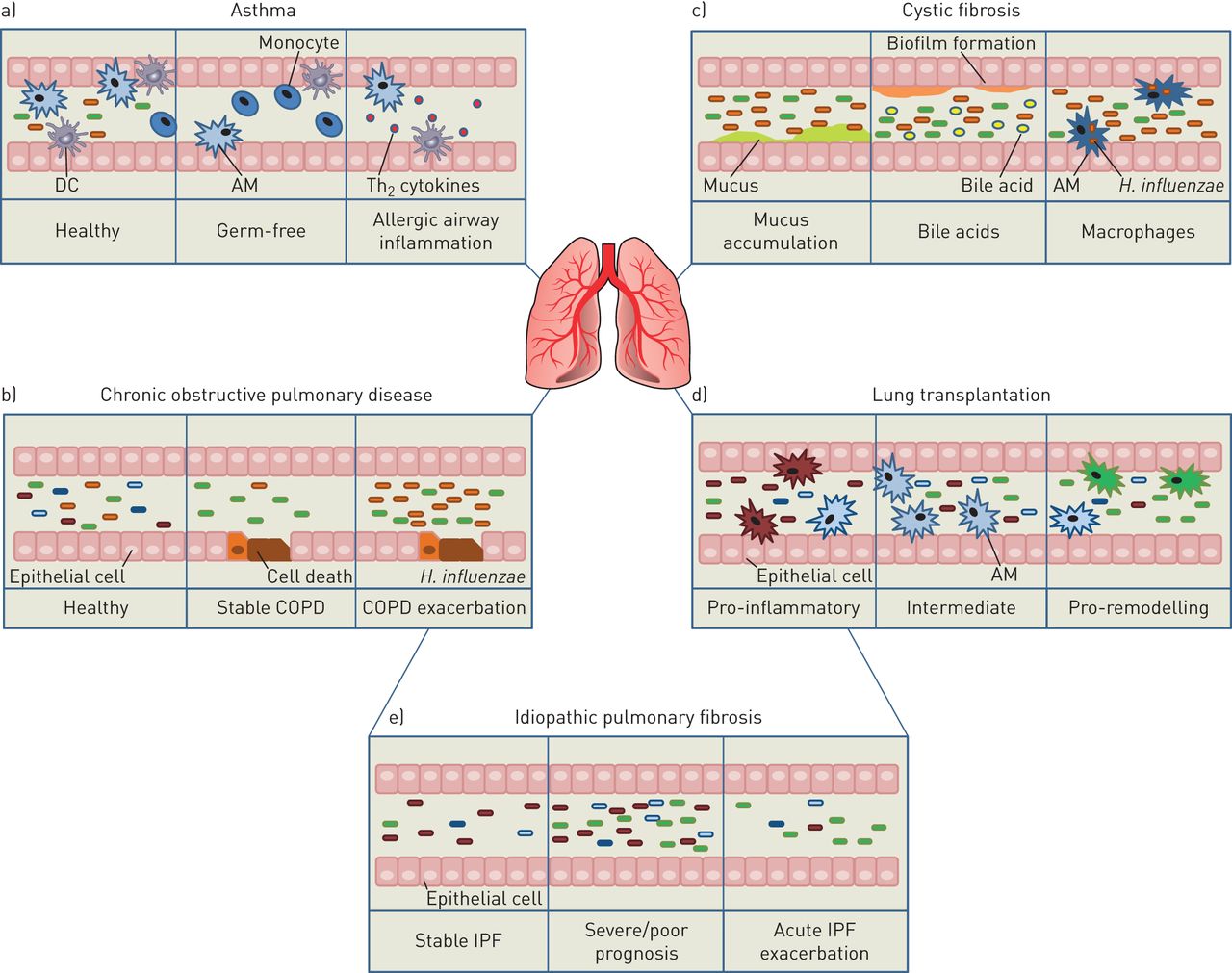
The Immunoecology Of The Respiratory Tract Microbiome
In the “Life in Antarctica” model of the lung microbiome, the balance of three factors determine and shape the lung microbiome : microbial immigration into the airways, elimination of microbes from the airways, and the relative reproduction rates of the microbes found in the airways, which is determined by the growth conditions of the region. Any change in the lung microbial communities, within an individual or as viewed across disease states, must be the result of a change in one of these three factors. Evidence from our lab and others strongly points to subclinical microaspiration as the primary source of microbial immigration, , , , , . Subclinical microaspiration of pharyngeal secretions among healthy subjects is well-documented. Other contributing factors can include the inhalation of bacteria from the air and direct migration along airway mucosal surfaces. Microbial elimination is accomplished by a combination of mucociliary clearance, cough, and host immune defenses .
Figure 2
Model of the environmental, microbial, structural, and immunologic factors that control the composition of the lung microbiome during health and disease.
Recommended Reading: What Does It Mean When You Keep Having Heartburn
Lung Microbiome And Copd
The prevalence of COPD is increasing, and its management is a formidable challenge to healthcare systems. There is growing evidence that the lung microbiome is altered during the course of the disease and contributes to COPD pathogenesis . Hilty et al., provided early evidence of the presence of dysbiosis in COPD. 16S rRNA analysis was performed on DNA collected from swabs from the nose and oropharynx, and bronchial brushings from the left upper lung lobe of COPD patients and healthy controls. Sequencing showed that COPD patients have a distinct microbiome in their lungs compared to healthy individuals. Specifically, pathogenic Proteobacteria were more common in COPD than Bacteroidetes, with Prevotella spp. being especially reduced . Subsequent studies showed the predominance of Firmicutes, Bacteriodetes, Proteobacteria, Fusobacteria, and Actinobacteria in healthy individuals, in contrast to pathogenic Haemophilus, Streptococcus, Klebsiella, Pseudomonas, and Moraxella in COPD patients . We propose that the lung microbiome is dynamic and transient, with specific taxa being present at different stages of disease progression and impacted by clinical factors such as age, smoking, medications, or seasonal changes .
The Lung Microbiota And Pneumonia: The Path Forward
Studies of the lung microbiome have shown promise in providing important insights into pneumonia pathogenesis. Adequately powered longitudinal lung microbiome studies are needed to describe transitions to and from homeostatic and dysbiotic states during the development and resolution of pneumonia. Such studies may require complex study designs sets and will be facilitated by research with the stated goal of identifying optimal and less invasive sampling methods for the lower respiratory tract. Additional work should also examine the virome and mycobiome with a focus on interkingdom interactions that influence progression to pneumonia and resolution of disease. The field is moving beyond 16S rRNA sequencing and into shotgun metagenomic sequencing and RNA profiling, to provide species- and strain-level resolution, to identify antimicrobial resistance genes, and to gain further insight into the functional capacity of the microbiota. Newer technologies that allow for examination of the proteome and metabolome may provide information regarding the host response. Use of these tools and technologies to expand the current body of lung microbiome research has the potential to reduce ineffective treatments and the overuse of antibiotics and may lead to better definitions of pneumonia and microbiome-based biomarkers for disease.
Read Also: Azo Probiotic For Bv
Gut Microbiome Impacts Immune Responses
Several mechanisms have been proposed to describe the impact of the gut microbiota on lung physiology. A prevailing hypothesis is that changes in gut microbial composition impact host immune responses in the lung. Support for this hypothesis demonstrating that the gut microbiome impacts immunity in the gut, lungs, and systemically, is discussed in the following section.
Microbiome As A Therapeutic Target In Lung Cancer
Given the role of microbiome in cancer development and progression, the modulation of microbiome can be used as a therapeutic strategy in lung cancer treatment. The concept of administration of microbial compounds to improve human health is not new, but the administration of microbes could in fact have beneficial effects . In order to modulate the microbiome, the use of pre-/probiotics has been suggested to synergize the effects of bacteria and substrates, promoting a health benefit and also supporting a healthy digestive tract . Manipulation of microbiota may also be useful in cancer prevention. A recent study demonstrated that the intake of a probiotic supplement containing Lactobacillus helveticus could decrease IL-17, reduce T cells, and suppress tumor formation, maybe due to gut microbiome alteration , as observed in Figure 1.
Read Also: How To Take Align Probiotic
Workshop Limitations And Further Reading
The present manuscript is a report of a workshop on which some aspects that deserve comment were not covered. Several investigators have addressed the role of the microbiome in asthma and paediatric diseases other than CF, a research field that has recently been reviewed . These reviews interestingly describe mouse and human data of the lunggut axis on asthma development. Similarly, the role of anaerobic bacteria in respiratory disease has been only marginally addressed in diseases such as CF and bronchiectasis to date and needs focused research.
Balancing The Bodys Bacteria
Dr. Tom Louie, MD
Clostidium difficile is painful hospital infection that causes severe fever and prolonged diarrhea, which can lead to blood poisoning and death. Treatment has greatly improved thanks to Dr. Louie’s groundbreaking work. He’s a pioneer in the use of fecal transplants to put the bodys bacteria back into balance to cure the infection. He also created a pill treatment that eliminates the uncomfortable process of transplanting feces into intestines.
Advancing knowledge of the bodys bacterial balance can lead to great changes. For example, clinical trials are proving that a probiotic pill given to at-risk adults admitted into the hospital can prevent nearly 75 percent from contracting C. difficile. Our study of the microbiome in Calgary patients taking probiotics is helping advance our knowledge of how to balance bacteria in the body.
Recommended Reading: Does Peanut Butter Cause Heartburn
S Rrna Gene Sequencing
16S rRNA gene sequencing is an easily applicable technology that gives a description of the bacteria present in a complex biological mixture, allowing investigation of whole communities and the identities of their constituent members.
Bacteria are traditionally classified taxonomically into phyla, genera and species based on differences in phenotypic characteristics, and phenotypic tests still underpin much of standard clinical microbiology. Genetic sequence variation provides a more-robust means of tracing the evolution of organisms from the most simple to the most complex. Carl Woese and others during the late 1970s pioneered sequencing of the small subunit ribosomal gene to address bacterial phylogeny. This gene was particularly suitable because ribosomes are a conserved component of the transcriptional apparatus of all DNA-based life forms.
The gene comprises nine constant regions and nine hypervariable regions . The variable regions enable sequence-specific discrimination between different bacteria. Using primers to span hypervariable regions, the coupling of 16S rRNA polymerase chain reaction with next-generation sequencing of amplicons makes it feasible to sequence many samples at low cost. The sequences amplified can be identified by mapping to reference databases, thereby providing semiquantitative estimates of the bacteria present in any sample. Careful PCR primer design is essential to control biases for and against particular groups of organism.
How Does The Interaction Between Intestinal Microbiota And The Immune System Affect Health And Diseases
The microbiota has been shown to be implicated in a range of immune-mediated disorders including inflammatory bowel disease, autoimmunity, and allergy. Therefore, altering the composition or function of the microbiota is a promising therapeutic approach to modulate immune responses involved in disease. Because T helper cells are involved in both immune adaptations to colonization and pathologic processes in disease, the lab focuses on the interaction of the microbiota with T helper cells.
The team uses germ free mice in combination with genetically modified commensal species to interrogate whether and how the microbiota can modulate T helper cell responses. This allows for carefully controlled and defined experiments to study how the microbiota modulates T helper cell response, but also how, in return, T helper cell responses directed at the microbiota impact on the microbiota at the level of transcriptional or metabolic activity and microbiota composition.
These are important parameters that need to be defined to be able to rationally design therapies that involve use of the microbiota as a tool to therapeutically modulate immune responses in disease.
Read Also: Can Prenatal Vitamins Cause Diarrhea
An Ecological Model Of Pneumonia
The recognition of an active resident lung microbiome during health calls into question the paradigm that pneumonia occurs when a virulent pathogen invades the normally sterile space in the lungs. The limits of this view have long been apparent in studies that have identified potentially pathogenic bacteria and viruses in the lungs of individuals without clinical signs of pneumonia , and from the lack of correlation between clinical cure and bacteriological cure in clinical studies . Ecological models of pneumonia, proposed by Dickson and colleagues , posit that pathogenesis involves a rapid shift from a homeostatic state in the lung microbiome toward a state of dysbiosis characterized by low microbial diversity, high microbial burden, and host inflammation. Processes that disrupt homeostasis include antibiotics, environmental exposures , mechanical ventilation, direct and indirect interactions between microbes, and other factors that influence the balance between immigration, emigration and microbial growth rate in the lungs. Pneumonia can be considered as a disease that exists on a continuum of microbiota abundance, diversity, and composition with a range of corresponding immune states. As a complex system, small changes can alter states and lead to a cycle of positive and negative feedback loops and then to decompensation in the lung microbiome .
What Is The Microbiomes Role In Immunity And Disease
Kathy McCoy, PhD
Dr. McCoy leads research in the 10,000-square-foot facility germ-free facility to understand the interaction between the microbiome and the immune system to prevent and treat illness. The facility houses thousands of mice under germ-free conditions. This lets researchers transplant specific microbiomes into germ-free animals. When we pair this with incredible imaging capacity, researchers can observe the behaviour of our microbiome in an environment free from other bacterial or viral contaminants. And how their introduction or removal shapes human health. That allows us to develop more personalized therapies, treatments and preventative measures for people living with chronic diseases.
International Microbiome Centre
You May Like: Cottage Cheese Probiotics
Lung Microbiota And Cancer
Table 1 Microorganisms found in lung cancer patients: clinical studiesFull size table
These data point out that bacteria in resected normal lung tissue might serve as biomarkers to predict disease recurrence risk in early-stage NSCLC. If the prognostic value of lung microbiota composition is validated in future studies, it will also represent a novel target for therapeutic intervention to improve RFS in lung cancer patients .
An association between the presence of specific bacteria and clinical outcome has been also reported in a cohort of 165 non-malignant lung tissue samples collected from lung cancer patients with different stage disease. The genus Thermus was more abundant in tissue from advanced stage patients , while Legionella was higher in patients who developed metastases . Again, the non-malignant lung tissue was characterized by higher microbiota alpha diversity than the matched tumor tissue, confirming the results above reported.
Another important aspect to be considered is that bacteria within the tumor microenvironment influence cancer cell growth by exerting immunomodulatory effects.
We previously observed that the aerosol delivery of various agents, such as TLR agonists and cytokines, was able to favor the immune-mediated control of experimental lung metastases, indicating the feasibility of influencing local lung immunity through a non-invasive strategy .