Nasal Bacterial Alpha Diversity And Asthma Activity
Rarefaction curves calculated based on Faith’s phylogenicdiversity for healthy controls, non-exacerbated asthma, and exacerbated asthmashowed a positive trend between nasal bacterial alpha diversity and asthmaactivity . These differences inphylogenic diversity were not statistically significant, however.
Trend toward increased nasal bacterial alpha diversity with greater asthmaactivity
Rarefaction plot showing the total branch length using Faith’s phylogenicdiversity is plotted against fraction of retained reads for healthycontrols and subjects with non-exacerbated asthma and exacerbated asthma . Differences were not statisticallysignificant at Pâ¤0.05.
Therapies Change The Urt Microbiome Composition And Diversity
Intranasal corticosteroids , saline rinses, antihistamines, and antibiotics are the current medical therapies of choice for inflammatory disorders of the upper respiratory tract . In contrast to anti-inflammatory substances that act through immunomodulatory mechanisms, antibiotics and some INS have antimicrobial properties and thus impact the microbial community directly .
The Respiratory Microbiome In Asthma
Chronic rhinosinusitis and asthma are both chronic diseases characterized by respiratory mucosal inflammation, often rich in eosinophils. Approximately half of CRS patients also suffer from asthma, asthmatics are much more likely to have CRS than the general population, and the two diseases share an association with allergic diseases. There are similarities in the deviations of the microbiome from control subjects in these diseases as well.
Colonization with Streptococcus pneumoniae, Haemophilus influenzae, Moraxella catarrhalis or a combination of these organisms in neonatal samples from lung is associated with increased risk of asthma later in life. This provides evidence of a persistent effect of microbial colonization of airways on initiation or progression of inflammatory respiratory disease. Along the same lines, multiple longitudinal studies have shown an association between viral respiratory illness with wheezing due to RSV and RV and subsequent development of asthma later in life.
Read Also: What Does It Mean When You Keep Having Heartburn
Microbiota Composition And The Integration Of Exogenous And Endogenous Signals In Reactive Nasal Inflammation
Vincenzo Casolaro
1Department of Medicine, Surgery and Dentistry, Scuola Medica Salernitana, University of Salerno, 84081 Baronissi , Italy
2Department of Neurosciences, Reproductive and Odontostomatological Sciences, University of Naples Federico II, 80131 Naples, Italy
Abstract
1. Introduction
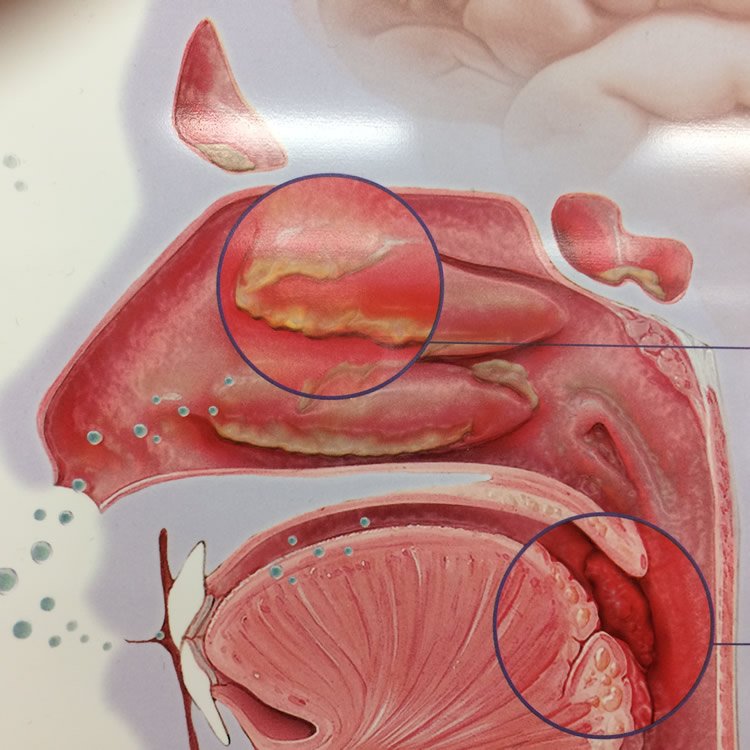
The nose, the uppermost portion of the respiratory tract, serves important physiologic functions, such as air filtration, warming, humidification, and olfaction. It consists of two cavities or fossae extending from the external nostrils to the choanae and separated longitudinally by an osteocartilaginous septum. The lateral wall of each fossa provides insertion to three turbinates, or conchae, which divide the cavity in three passages, or meatuses . These anatomical structures are essential to the air conditioning functions of the nose in that they expand the surface exposed to inhaled air. While the anterior nares and vestibule are lined with a skin-like stratified, keratinized epithelium, the nasal fossa proper is entirely coated with respiratory mucosa, consisting of a ciliated, highly vascularized, pseudostratified epithelium containing a sizeable number of mucus-producing goblet cells. The extensive vascularization of the nasal mucosa favors its air warming and humidifying functions, whereas the sticky seromucous secretions contribute to air filtering by effectively trapping inhaled particulate matter .
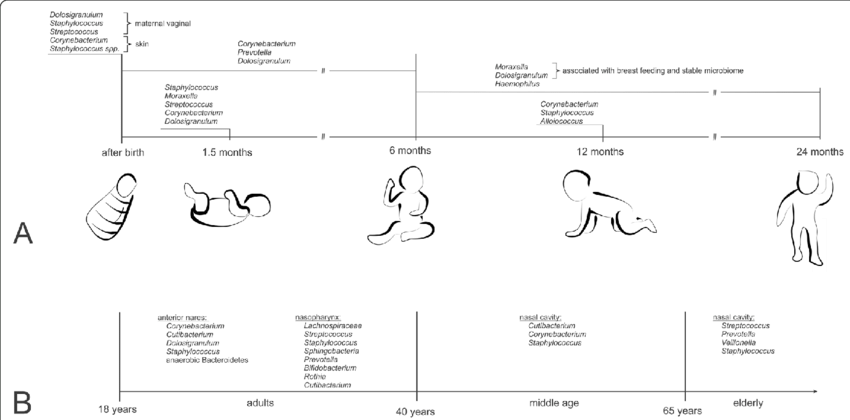
2. Towards the Definition of a Healthy Nasal Microbiota
Probiotic Intervention In Respiratory Tract Infections
Given the significant role of the nose and throat microbiome in the susceptibility to influenza virus infection,,, there has been some investigation into the potential role of probiotics in the modulation of the nasal microbiome and innate and adaptive host immune responses. Salk et;al. measured changes to the nasal microbiome in young adults who received intranasal administration of live attenuated influenza vaccine and found that LAIV altered the nasal microbiome, with increased taxa richness and variations in influenza-specific IgA antibody production.
Furthermore, molecular profiling studies have recently revealed that species-specific interactions play an important role in avoiding S. aureus nasal colonization and persistence. Specifically, the Corynebacterium genus interacts with S. aureus in the nasal cavity and artificial inoculation of Corynebacterium pseudodiphtheriticum into the nasal cavity appears to eradicate S. aureus nasal colonization. Hardy et;al. determined that this is because C. pseudodiphtheriticum selectively targets S. aureus for killing, mainly through Agr QS, its primary virulence factor. This may confer selective advantage to S. aureus strains deficient in Agr QS, inducing a switch from a pathogenic state to a commensal state. Further studies are needed to characterize the therapeutic use of C. pseudodiphtheriticum-derived factors as bactericidal agents against S. aureus.
Don’t Miss: Can Align Probiotic Cause Nausea
How Do I Get Rid Of Bad Bacteria In The Gut
There are actually relatively few bacteria that you should get rid of. While somelike salmonella or campylobacterare very bad news, bacteria are mostly neither good nor bad: at normal levels, theyre neutral. In an unbalanced microbiome though, some microbes can multiply to abnormal levels.;
A good gut health test will show you which microbes have multiplied beyond healthy levels. Again, its all about diversity and balance. Read more about how getting rid of overgrown bacteria is not always a good plan in our blog How to get rid of bad bacteria in the gut.
What Is The Role Of Nasal Microbiota In Respiratory Recurrent Infections
Prof. Desiderio Passali, from University of Siena, Italy will present his studies on the Role of nasal Microbiota in respiratory recurrent infections during the 5th Targeting Microbiota World Congress 2017.
According to Prof. Passali, the presence of a normal naso-sinusal microbial ecosystem, the so called nasal microbiota, is the mainstay for a good homeostasis of respiratory tract.
An imbalance of the nasal microbiota can lead to upper respiratory recurrent infections, which represent the most common illnesses in the patient outsetting.
Special interest grow up in the last years on possible approaches to restore the nasal microbiota. In this context, the topical administration of bacterial species belonging to the healthy human nasal microbiota can offers great benefits for the patient, contributing to the re-colonization process, re-establishing microbial balance, and reducing the level of potential pathogens and could represent a valid alternative to antibiotic.
In particular, the combination of two -hemolytic streptococci, S. salivarius 24SMB and S. oralis 89a, was able to interfere, in vitro, with the biofilm formation capacity of pathogenic strains of the upper airway; moreover, this combination, resulted able, in various clinical trials, to significantly reduce the recurrence rates of upper respiratory infections.
For more information: www.targeting-microbiota.com
Also Check: Can Soy Milk Cause Diarrhea
The Nasal Microbiota Of Dairy Farmers Is More Complex Than Oral Microbiota Reflects Occupational Exposure And Provides Competition For Staphylococci
- Sanjay K. Shukla ,
Contributed equally to this work with: Sanjay K. Shukla, Zhan Ye, Matthew Keifer
Roles Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Project administration, Resources, Supervision, Validation, Visualization, Writing original draft
* E-mail:
Affiliation Molecular Microbiology Laboratory, Center for Human Genetics, Marshfield Clinic Research Institute, Marshfield, Wisconsin, United States of America
-
Contributed equally to this work with: Sanjay K. Shukla, Zhan Ye, Matthew Keifer
Roles Formal analysis, Methodology
Affiliation Biomedical Informatics Research Center, Marshfield Clinic Research Institute, Marshfield, Wisconsin, United States of America
-
Affiliation National Farm Medicine Center, Marshfield Clinic Research Institute, Marshfield, Wisconsin, United States of America
-
Affiliation National Farm Medicine Center, Marshfield Clinic Research Institute, Marshfield, Wisconsin, United States of America
-
Affiliation Division of Laboratory Medicine, Marshfield Clinic, Marshfield, Wisconsin, United States of America
-
Contributed equally to this work with: Sanjay K. Shukla, Zhan Ye, Matthew Keifer
Roles Conceptualization, Data curation, Funding acquisition, Investigation, Project administration, Writing review & editing
Affiliation VA Puget Sound, Seattle, Washington, United States of America
Knowledge Gaps Conclusion And Outlook
Determining whether the detected changes or dysbioses in the URT microbiome associated with disease are markers or drivers presents a major challenge. There has already been some progress towards identifying biomarkers that could be used for early diagnosis of URTIs, such as Microbacterium spp., Streptococcus spp. or Faecalibacterium spp., whereas identifying targets for microbiome-based therapies remains more difficult. The ability to sample from disease-relevant sites within the URT is helpful in this regard, as it enables the identification of microbial candidate disease drivers whose abundance is positively correlated with both the site and incidence of disease, while negative correlations reported from the disease site are similarly more likely to be relevant, pointing to a possible protective role that might be harnessed in probiotic therapy. It will therefore be important to address the methodological challenges of sampling from less accessible URT sites, and to continue to develop appropriate sampling tools to minimise contamination from neighbouring sites. Further investigation of the co-operative and competitive interactions of microbes and host may also be helpful in guiding rational choices in the pursuit of causal connections and therapeutic goals. However, establishing causality and demonstrating the efficacy of proposed treatments requires other approaches, such as animal models and clinical trials.
Read Also: Does All Cottage Cheese Have Probiotics
Microbiota Variation Among Different Patients
FIGURE 2. Non-metric multidimensional scaling plot displaying 16S rRNA gene-based bacterial community composition at bacterial genus level. Subjects with CRS are signified by red symbols, with healthy subjects in blue symbols. Each subject is represented by six different samples, as outlined for Figure 1. Sequence data were compared among samples using BrayCurtis similarity.
Host Factors And The Microbiome In Crs
The interplay between the innate and adaptive immune systems, the microbial communities and inflammation in sinonasal mucosa is rather complex and not completely understood yet. Th2 inflammation in eosinophilic CRS has been associated with down regulation of elements of innate immunity. Reductions in antimicrobial peptides like human beta-defensin 2 and surfactant protein-A and decreased expression of TLR9 have been described in CRS. These alterations can influence the defense against microbial pathogens and affect microbial colonization. On the other hand, colonization with fungi and specific bacteria like S. aureus has been linked to maladaptive Th2 responses. Microbes and their products are ligands for the innate immune receptors that are also present on inflammatory granulocytes like basophils and mast cells that are found in CRSwNP. Activation of these cells through these receptors could result in recruitment of other inflammatory cells involved in Th2 response and progression of the inflammation in CRS.
In the following paragraphs, we have briefly summarized the findings regarding changes in the mucosal immune system in CRS that can influence the microbial communities in the sinonasal cavity.
S100 proteins are involved in epithelial defense and repair, and have antimicrobial functions. A few studies have shown a significant reduction in S100 proteins including S100A7 and S100A8/A9 in CRSsNP and CRSwNP , which can lead to a diminished innate immune response.
You May Like: Azo Probiotic For Bv
How The Sinus & Gut Microbiome Relate
The gut is often referred to as the bodys second brain, which implies that keeping a healthy gut is extremely important for our health and our immune system. The stronger our immune system is, the more chances we have to fight off viral infections that can result in sinusitis. Again, the gut microbiome is home to hundreds of microbial species that perform a wide range of tasks to maintain normal immune and digestive functions.
According to scientific evidence, chronic diseases, inflammation, and even the formation of cancer are associated with the presence of certain bacteria that live in the gut. At the same time, other microbes that also live in the intestines are health-promoting and disease-preventing.
Luckily, we can control which microbes live and thrive in our gut through our food and lifestyle choices.
Unfortunately, the Western lifestyle is characterized by diets that include a plethora of unhealthy processed foods, fast foods, lots of red meat, foods low in fiber, sweets, and the frequent use of antibiotics, alongside lack of sufficient sleep, no exercise, too much alcohol, and smoking. This way of life does not promote a balanced microbiome and feeds the harmful bacteria. Over time the beneficial bacteria get depleted, allowing the harmful ones to take over and cause a variety of diseases, including respiratory health conditions that lead to sinusitis.;
Your Nose Has Its Own Microbiome And It Might Be Possible To Change It Study Shows
It’s been widely reported how the mix of our gut bacteria can end up having a direct impact on our health. Now,;new research points to a similar link between our wellbeing and the microbiome in the nose.
That’s right your nose is home to an entire host of microorganisms, just like the rest of your body. The different types of ‘good’ or ‘bad’ bacteria specific to the nasal cavities might end up protecting you from certain health issues, or putting you more at risk of others.
The inspiration for this new study came from the mother of one of the researchers, who was having problems with headaches and chronic rhinosinusitis a condition that causes pain and swelling in the sinuses and nasal cavity.
“My mother had tried many different treatments, but none worked,” says microbiologist Sarah Lebeer, from the University of Antwerp in Belgium. “I was thinking it’s a pity that I could not advise her some good bacteria or probiotics for the nose. No one had ever really studied it.”
Lebeer and her team analysed nose bacteria from 100 healthy volunteers and 225 people with chronic rhinosinusitis, characterising 30 different families of bacteria in all. One family in particular stood out: lactobacilli.
The next problem for the researchers was testing whether the presence of these lactobacilli could be artificially introduced as a probiotic something that can’t be easily done using an animal model.
The research has been published in Cell Reports.
Also Check: Why Does Lettuce Give Me Diarrhea
The Fungal Microbiome In Sinusitis
Study of nonbacterial microbiota in the nose has been an area of interest in recent years. The association of fungi with CRS has been investigated in multiple studies, with controversial results. In the late 1990s, fungal cultures identified colonization in more than 90% of CRS patients and drew significant attention to this field. In this way, several fungal species were identified in CRS. But further studies showed evidence of fungal colonization in the nose in more than 90% of healthy controls from the same population . Interestingly, studies in other populations showed a significantly lower level of fungal colonization in CRS. Similar fungal culture techniques yielded positive results in 13% of a Turkish and 36% of a Malaysian CRS series. Fungal organisms have also been found in biofilm formations in CRS . Studies using antifungal treatments in CRS have primarily failed to support a clinical use of this approach.
Viral Microbiome And Crs
A majority of CRS exacerbations occur in seasons with a high prevalence of viral infections . Using multiplex PCR for respiratory viruses, Cho et al found viral nucleic acid sequences in 64% of sinus scrapings and 50% of nasal lavage samples in the CRS group, which was significantly higher than controls . In their study, rhinovirus was the most frequently detected virus. In a study from China, test of the scraped epithelial cells from the middle meatus revealed an evidence for respiratory viruses in 68.66%, 73.77% and 75.47% of CRSwNP, CRSsNP and control cases, which was not statistically different. In 2006 Jang et al showed that 21% of turbinate epithelial cell samples from CRS patients were positive for rhinovirus detected by reverse-transcription PCR. In that study, RV was not detected in the lavage of any of these patients. In contrast, Wood et al did not find any evidence for respiratory viruses in 13 CRS patients tested by PCR for common respiratory viruses including, but not limited to, rhinovirus, parainfluenza viruses 1, 2, and 3, and respiratory syncytial virus .
The role of rhinovirus infection in asthma exacerbation has been well established and the similarities between the two diseases suggest a role for viral infection in triggering exacerbations of CRS, a hypothesis that calls for further investigation. However, current literature in this field is not conclusive.
Don’t Miss: Does Oikos Have Probiotics
Microbiota Variation Among Different Sites Within One Patient
Bacterial community composition and inferred abundance did not vary in a predictable manner across the six assayed sites within one patient . As indicated above, the microbiotas of CRS patients were particularly variable, even within some individuals. Only for patients 4, 6, and 9 were the communities relatively consistent for the six sample sites, in two cases dominated by Corynebacterium and in the third by Staphylococcus. Among the other diseased individuals, the microbiota of patient 1 was comprised almost exclusively of the gammaproteobacterium Moraxella, except for one nostril in which the anterior nares also contained a considerable amount of Corynebacterium. In contrast, bacterial communities at the six sites among the six healthy individuals remained fairly consistent at phylum and genus level. The bacterial load in patient 1 was significantly greater compared with those of the other five healthy individuals, but no obvious pattern was observed for any individual site within the nasal cavity. Bacterial diversity did not differ significantly between sites for CRS and healthy individuals. The same applied to comparisons between nostrils.
S Rrna Gene Amplicon Pyrosequencing
Bacterial 16S rRNA gene fragments from the extracted genomic DNA were amplified using primers 347f and 803r, which have been used previously to characterize the bacterial community of the human foregut . The applicability of these primers to sinus microbial communities was validated in silico by using Probe Match in the RDP database and SILVA databases. Sample preparation for amplicon pyrosequencing was as described previously , with some minor modifications. In brief, the aforementioned 16S rRNA gene-targeting primers, complete with pyrosequencing adaptors and unique multiplex identifiers on the forward primer, were used in equimolar concentrations together with dNTPs , PCR buffer , MgSO4 , 0.5U Platinum Hi-fidelity Taq and PCR-certified water to a final volume of 25 L. PCR amplification was performed in an Applied Biosystems Mastercycle gradient PCR machine with an initial denaturing step of 94°C for 3 min, followed by 35 cycles of denaturation , annealing , and elongation , with a final elongation step at 70°C for 3 min. Amplified products were purified using Agencourt AMPure beads , quantified using Picogreen, and qualitatively checked on Agilent 1200 Bioanalyzer DNA 1000 chips . A nested-PCR approach, using primers 616V and 1492R for the initial amplification, was adopted for the samples of two CRS patient samples , as the original PCR did not yield any products.
Read Also: What Does Ibd Mean In Text