Composition Of The Oral Microbiome
A wide range of microorganisms are present in the oral cavity. It is in constant contact with and has been shown to be vulnerable to the effects of the environment.
The human microbiome consists of a core microbiome and a variable microbiome. The core microbiome consists of predominant species that exist at different sites of the body under healthy conditions. The variable microbiome has evolved in response to unique lifestyle and genotypic determinants and is exclusive to an individual.
The microbial ecology of the oral cavity is complex and is a rich biological setting with distinctive niches, which provide a unique environment for the colonization of the microbes. These niches include the gingival sulcus, the tongue, the cheek, the hard and soft palates, the floor of the mouth, the throat, the saliva and the teeth.
Different surfaces in the mouth are colonized preferentially by the oral bacteria due to specific adhesins on their surface which bind to complementary receptors on an oral surface.
The normal microbiome is formed by bacteria, fungi, viruses, archaea and protozoa. The reports on a normal microbiome, however, are restricted to the bacteriome, and there are very few reports on the mycobiomefungal microbiome.
Oral cavity is one of the most well-studied microbiomes till date with a total of 392 taxa that have at least one reference genome and the total genomes across the oral cavity approaching 1500.
Gram positive:
Gram negative:
Onset Of Inflammatory Bowel Disease
The Inflammatory Bowel Disease Multi’omics Data team was a multi-institution group of researchers focused on understanding how the gut microbiome changes longitudinally in adults and children suffering from IBD. IBD is an inflammatory autoimmune disorder that manifests as either Crohn’s disease or ulcerative colitis and affects about one million Americans. Research participants included cohorts from Massachusetts General Hospital, Emory University Hospital/Cincinnati Children’s Hospital, and Cedars-Sinai Medical Center.
Alberta Bloom Study: Begin A Life Of Health With Observation And Optimization Of The Microbiome
The microbiome is inherited largely from our mother at birth and in our first years of life, forming an integral part of our unique physiological identity.
Recent studies have found that changes in the microbiome very early in life can herald the onset of disease years later, but more research is needed. New data also suggests the maternal microbiome could have significant effect on a childs health. Understanding how the maternal and early life microbiome interacts with our human genetic background and environment is one of the greatest frontiers in modern medicine.
The BLOOM study looks at the microbiomes of 1,000 babies born in Alberta. We’ll examine how the microbiome changes from the womb, through birth, and through the use of antibiotics in the first five years of life.
This study aims to trace the microbial links between mothers and their neonates in pregnancy, birth, and up to one year postpartum. The broad goals of the study: Molecular surveys of the microbiome and host to identify biomarkers, along with their mechanistic underpinnings. This will provide diagnostic value or predictive information of long-term health outcomes.
Don’t Miss: Does Peanut Butter Cause Heartburn
Normal Microbiota As Host Defense
The human microbiome is now recognized as a major host defense against bacterial pathogens by providing colonization resistance, maintaining a balance of commensals to pathogens, and by priming the immune system .5 Altering or disrupting the normal microbiota by antibiotics facilitates the expansion of enteric pathogens as Clostridium difficile and Salmonella typhimurium or selection of antibiotic-resistant members of the microbiome. Similarly, changes in human physiology, for example, exposure of skin to elevated temperatures and humidity, chronic stress, host immune suppression, or active behavioral changes, such as smoking, can cause a commensal-to-pathogen switch. Recent studies have demonstrated that certain resident microbiota can resist pathogen colonization and infection. For example, matched volunteers were inoculated with Haemophilus ducreyi into the arms, and the subsequent infection either resolved or resulted in formation of abscesses characterization of the skin microbiome before, during, and after the experimental inoculation showed that the microbiomes of those with pustule formation and of those with resolved infection were distinct and influenced the course of the H. ducreyi infection.6
Michael J. Orlich, ⦠Sarah Jung, in, 2017
Development Of Innovative Microbiome

Though this field is still in its early days, development of a number of microbiome-based disease preventions and interventions are already underway, some of which are being evaluated by the FDA. Fecal microbiota transplantation shows efficacy for recurrent C. difficile gut infections but has not yet shown reproducible efficacy for other conditions. Further, FMT is acknowledged as a black box approach for treating disease so more precise strategies have been under development. These strategies currently fall into four general categories: host microbiota-derived live biotherapeutic products for reducing inflammation, restoring gut barrier function, or, in some cases, improving colonization resistance against pathogens treatments which target-specific pathogens using microbiome-sourced antibiotics that can target antibiotic-resistant pathogens as well as the use of narrow host-range phage prebiotics which are microbial growth substrates that can stimulate the metabolism and growth of specific commensal microorganisms and specialized metabolites or bacterial cellular components which can regulate or limit specific microorganisms or act directly on host pathways. A few examples of these promising strategies developed during the portfolio analysis period will be noted here.
Recommended Reading: Cabbage And Ibs
About The Human Microbiome
The Human Microbiome is the collection of all the microorganisms living in association with the human body. These communities include eukaryotes, archaea, bacteria and viruses. Bacteria in an average human body number ten times more than human cells, for a total of about 1000 more genes than are present in the human genome. Because of their small size, however, microorganisms make up only about 1 to 3 percent of our body mass . These microbes are generally not harmful to us, in fact they are essential for maintaining health. For example, they produce certain vitamins that we do not have the genes to make, break down our food to extract nutrients we need to survive, teach our immune systems how to recognize dangerous invaders and even produce helpful anti-inflammatory compounds that fight off other disease-causing microbes. An ever-growing number of studies have demonstrated that changes in the composition of our microbiomes correlate with numerous disease states, raising the possibility that manipulation of these communities could be used to treat disease.
New Approaches To Study The Role Of Host Genetics In The Microbiome
More attention should be paid to the role of host genetics in microbiome assembly in host-microbiota interactions and in disease. There remains debate in the literature with some studies suggesting that host genetics plays a major role in the assembly of the infant gut microbiome with the environment playing a greater role in adult subjects so that the microbiome is considered a partially heritable trait. On the other hand, some studies have demonstrated interactions between host genetics, some members of the microbiome and factors such as diet, innate immunity, vitamin D receptors, as well as some autoimmune diseases. New approaches are needed to establish the conditions under which host genetics plays a role in microbiome structure and function as these kinds of data would be useful, for example, for stratifying subjects in cohort studies of disease risk or for testing treatments or interventions.
Don’t Miss: Antacid Fruit
Digestion And Food Breakdown
Until recently scientists believed that the human body was fully responsible for digestion of food but it turns out the bacteria in our gut plays a large role in what we are able to eat and digest. Bacterial enzymes in our gut can break down complex sugar other food molecules into fuel. These bacteria can also produce vitamins in the body, making them more essential than previously realized.
What Is The Human Microbiome Project
Worldwide research initiatives are mapping the human microbiome, giving insight into uncharted species and genes. One initiative is the Human Microbiome Project sponsored by the National Human Genome Research Institute , part of the National Institutes of Health . The HMP began in 2008 as an extension of the Human Genome Project. It is a 5-year feasibility study with a budget of $150 million and is being carried out in a number of centers around the US.
The purpose of the HMP is to study the human as a supraorganism composed of non-human and human cells, with the goal of describing the human microbiome and analyzing its role in human health and disease.
A major goal of the HMP is to characterize tthe metagenome of the microbiomes of 300 healthy people, over time. Five body areas are sampled: Skin, mouth, nose, colon, and vagina.
Read Also: Salad Gives Me Gas
The Role Of The Human Microbiota
Most members of the human microbiota benefit humans by providing them with traits that they would not otherwise possess. Some microorganisms found in the human gut, for instance, obtain nutrients from ingested food in return for assisting with the breakdown of food or preventing the colonization of the gut by harmful bacteria. There are, however, many microorganisms in the human microbiota that are closely related to pathogenic organisms or are themselves capable of becoming pathogenic. Examples include bacterial species of the genera Staphylococcus, Streptococcus, Enterococcus, Klebsiella, Enterobacter, and Neisseria.
Scientists studying obesity have detected an increased abundance of Prevotella and Firmicutes bacteria and of methanogenic archaea in obese individuals relative to normal-weight persons and persons who have undergone gastric bypass surgery. Scientists suspect that these microorganisms are more efficient at harvesting carbohydrates from food than are the types of microorganisms that dominate the gut flora of normal-weight individuals. The extra nutrients are then stored in the body as fat.
You May Like: Are Bananas Gassy
Considerations In Sampling The Human Microbiome
The first step in a microbiome study typically involves the collection of stabilized microbial biomass specimens that will be used and analyzed in various assays. Each sampling method for human-associated microbial community types has strengths and weaknesses that are driven by the dramatically different microbial ecologies in or on the body. The methods that have been established for gathering a sample of sufficient biomass for each major body site are described here, and limitations of each approach are noted.
Suggested Citation:Environmental Chemicals, the Human Microbiome, and Health Risk: A Research Strategy
the presence of human cells in the sample. Other sample types, such as mucosal brushing or rectal swabs, are also possible but are less well studied with respect to protocol consistency and community representation .
All human microbiome sampling protocols are sensitive to batch effectstechnical, not biologic, differences that arise from many stages of the sampling and data-generation process . Such effects can make data from multiple studies difficult to compare and, in the worst case, can introduce subtle differences that result in misleading conclusions. Gross differences in population structure, geography, or environmental conditions can change measured microbial communities. Differences in how samples are collected and processed can strongly influence microbiome
Suggested Citation:
Recommended Reading: Salads For Ibs
Don’t Miss: Can Align Probiotic Cause Nausea
How Will I Benefit From Participating In The Study
You may experience an improvement in the symptoms of fibromyalgia following the dietary intervention. If this happens, you will be able to continue following the diet after the study ends. If, following our analysis, we find that one diet is more efficient than the other, all participants who did not receive the more efficient diet will be invited to receive dietary counseling on it. This counseling will be offered by the study nutritionist team at no cost.
Brief Introduction To Early Period Of Human Microbiome Research Field
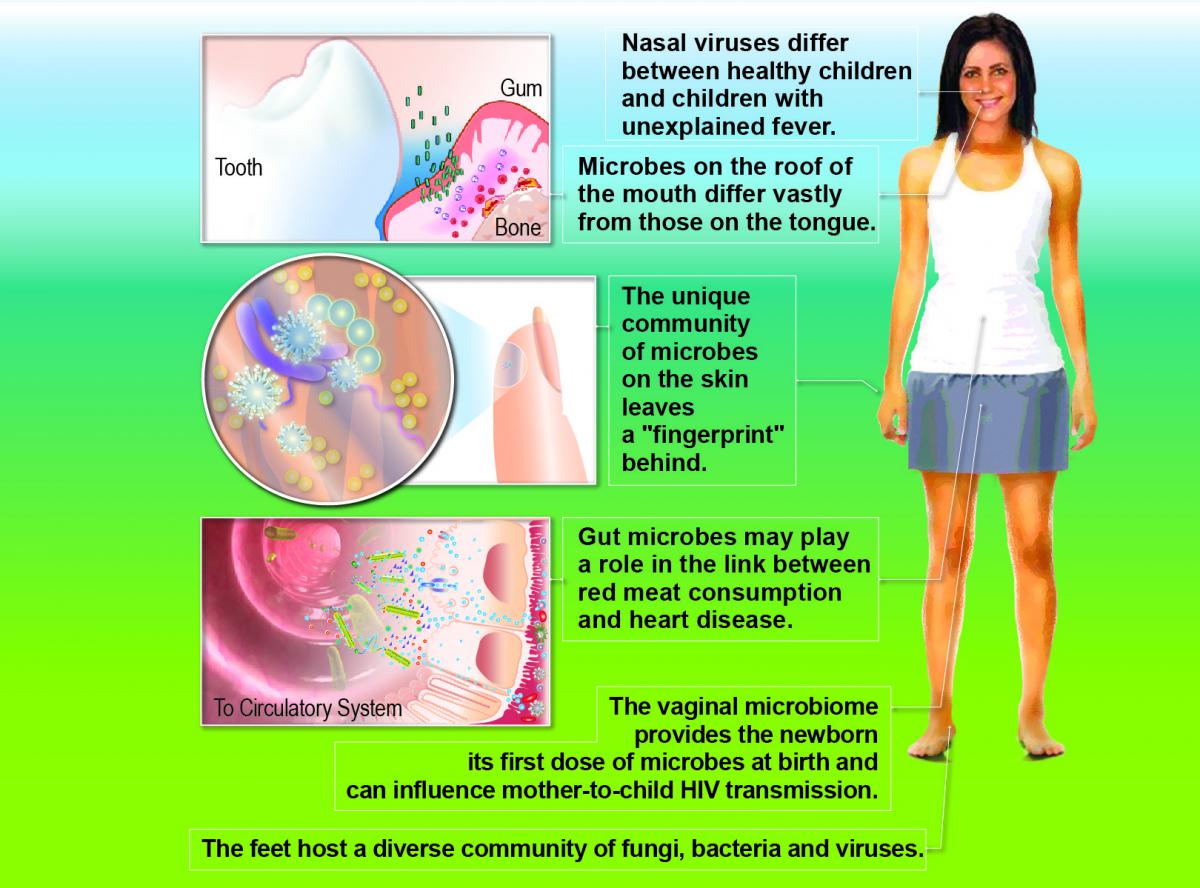
A number of pioneering studies, particularly in the early 2000s, helped lay the foundation for this field and inform the development of the HMP program. Though by no means exhaustive, examples include the extensive work on the diversity of oral pathogenic and nonpathogenic bacteria using 16S rRNA gene sequence analysis . This work later led to the development of a major resource for the field, the Human Oral Microbiome Database .
New approaches to the study of the gut microbiome led to seminal contributions in our understanding of the role of the gut microbiome in human biology, e.g., and in disease, e.g., . The germ-free humanized mouse model was developed during this early period and became a broadly adopted model for the study of the gut microbiome . Early work also led to foundational knowledge on the role of birth mode, e.g., and breast milk, e.g., in the development of the infant gut microbiome. Another important development was the recognition that some host disease phenotypes were associated with the gut microbiome, a property which could be transferred when the gut microbiome was transferred from the diseased donor to a recipient host, e.g., obesity .
Read Also: All Of The Following Are Common Causes Of Heartburn Except
Framework Of Sequence Data: Cohort Type And Data Type
One way of organizing the metagenomic sequence data generated by HMP1 is to split it by cohort type and data type.
Two primary cohort types:
Three primary data types:
No approval is necessary to access these metadata fields.
The Immune System And The Microbiome
Paul Kubes, PhD
Imaging is used to visualize both the microbiome and the immune system or immunobiome and investigate how these two systems interact. His latest work is examining how the gut microbiome develops in the presence and absence on antibiotics in neonate. And how the lung microbiome differs from other microbiomes to directly affect the lung immune system. In both cases, imaging is used to visualize the biology of immune cells.
You May Like: Aspirin Cause Constipation
Human Postmortem Microbiome Project
Figure 6.2. The Human Postmortem Microbiome Project will cultivate forensic and criminal investigative tools and databases for the law enforcement and forensic research communities. The HPMP will be the largest catalogue of sequencing and bioinformatics data of relevant microorganisms associated with internal organs, blood, skin, etc. and cadaver soil associated with human decomposition.
Future Areas Of Research
The microbiome is a living dynamic environment where the relative abundance of species may fluctuate daily, weekly, and monthly depending on diet, medication, exercise, and a host of other environmental exposures. However, scientists are still in the early stages of understanding the microbiomes broad role in health and the extent of problems that can occur from an interruption in the normal interactions between the microbiome and its host.
Some current research topics:
- How the microbiome and their metabolites influence human health and disease.
- What factors influence the framework and balance of ones microbiome.
- The development of probiotics as a functional food and addressing regulatory issues.
Specific areas of interest:
- Factors that affect the microbiome of pregnant women, infants, and the pediatric population.
- Manipulating microbes to resist disease and respond better to treatments.
- Differences in the microbiome between healthy individuals and those with chronic disease such as diabetes, gastrointestinal diseases, obesity, cancers, and cardiovascular disease.
- Developing diagnostic biomarkers from the microbiome to identify diseases before they develop.
- Alteration of the microbiome through transplantation of microbes between individuals .
You May Like: What Does It Mean When You Keep Having Heartburn
The Nih Human Microbiome Project
The Human Microbiome: How It Works + A Diet For Gut Health
Most people think of bacteria within the body as a cause of getting sick or developing certain diseases, but did you know that at all times there are actually billions of beneficial bacteria present within all of us? In fact, bacteria make up our microbiome, an integral internal ecosystem that benefits our gut health and the immune system.
Recently, the scientific community has really come to embrace the important role that bacteria have in fostering a strong immune system and keeping us healthy. Not only are all bacteria not detrimental to our health, but some are actually crucial for boosting immunity, keeping our digestive systems running smoothly, our hormone levels balanced and our brains working properly.
So what is the microbiome, why is it so important and how can we protect it? Lets find out.
Also Check: Cottage Cheese Probiotics